Key takeaway: Most probiotics fail not because of quality issues, but because the strains are incompatible with your individual gut microbiome. Colonisation resistance, the mechanism by which your existing bacteria reject newcomers, is personalised. A probiotic that works for one person may do nothing for another. Metagenomic testing can identify which strains will actually colonise your gut before you take them.
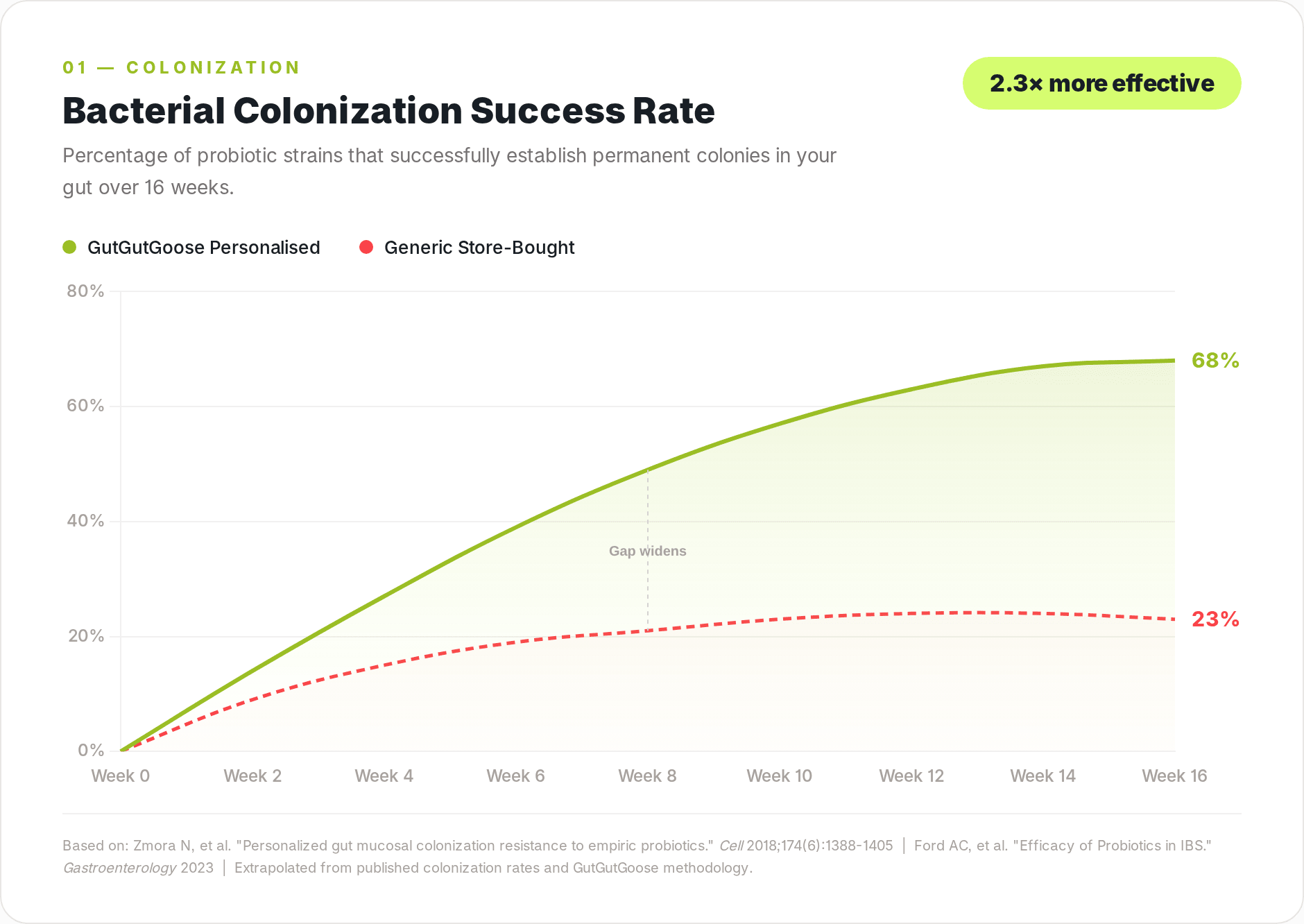
Most probiotics sold in Australia do not colonise the human gut. A 2018 study published in Cell by Zmora et al. at the Weizmann Institute of Science tracked 11 probiotic strains through participants' gastrointestinal tracts using endoscopic sampling and shotgun metagenomic sequencing. The researchers found that identical probiotic products colonised some participants' guts but passed through others with zero measurable mucosal effect. The determining factor was not the probiotic formulation. It was the individual's existing microbiome composition.
This finding has significant implications for the estimated 4.8 million Australians who take probiotic supplements annually. If colonisation depends on individual gut ecology rather than probiotic strain selection alone, then a one-size-fits-all approach to probiotics is fundamentally limited.
What Colonisation Actually Means
Colonisation refers to probiotic bacteria establishing viable, reproducing populations within the gut mucosa, as opposed to transiting through the digestive tract without adhering to or integrating with the existing microbial community. The distinction matters because transient bacteria, even if alive at the point of ingestion, provide limited long-term benefit once supplementation stops.
Goodoory et al. (Gastroenterology, 2023) conducted a systematic review and meta-analysis of 82 randomised controlled trials involving over 10,000 participants and found that while probiotics as a group showed modest benefits for conditions including irritable bowel syndrome, the effect sizes varied enormously between studies. The authors noted that individual variation in response was a major limitation across the literature.
The Zmora et al. (2018) data showed that in colonised mice, probiotics encountered marked mucosal colonisation resistance, and in human participants the majority of supplemented strains passed through the gut without establishing lasting populations. The probiotic strains survived manufacturing, survived stomach acid, and arrived in the colon alive, but the existing microbial community rejected them through a process called colonisation resistance.
How Colonisation Resistance Works
Colonisation resistance is the mechanism by which an established gut microbiome prevents new bacterial strains from taking hold. It operates through three primary pathways:
The Zmora et al. (2018) study demonstrated this directly: participants whose existing microbiomes were rich in species closely related to the probiotic strains showed higher colonisation rates. Participants whose microbiomes lacked those ecological niches rejected the same strains entirely.
Why the Same Probiotic Works for Some People and Not Others
The variation in probiotic response traces back to the composition of each individual's gut microbiome. Research published by Rothschild et al. (Nature, 2018) analysed 1,046 participants and found that environmental factors, including diet, medication use, and lifestyle, explained the majority of microbiome variation between individuals, with genetics accounting for only 2-8%.
This means two Australians eating similar diets, living in the same city, and taking the same probiotic supplement may have fundamentally different gut ecosystems. One person's gut may contain the ecological niche that a Lactobacillus rhamnosus GG strain needs to establish. The other person's gut may already be saturated with competing Lactobacillus species, leaving no available niche.
The practical result is what many consumers experience: a probiotic that a friend recommended with genuine enthusiasm produces no noticeable effect. This is not a placebo failure or a quality issue with the product. It is a mismatch between the probiotic strains and the individual's microbial ecology.
Common signs your probiotic is not colonising
The Dose Problem
Beyond strain mismatch, many commercial probiotics contain insufficient colony-forming units (CFU) to overcome colonisation resistance. A 2023 review in the World Journal of Gastroenterology noted that most clinical trials demonstrating probiotic efficacy used doses of 10 billion to 100 billion CFU per day, yet many retail products contain 1 billion to 5 billion CFU.
Even when a product label states a high CFU count, that number reflects viable organisms at the time of manufacture. Storage conditions, transport, and shelf life reduce viability. By the time a consumer ingests the product, the actual delivered dose may be a fraction of the label claim.
Therapeutic-dose probiotics, compounded to deliver a minimum of 50 billion CFU at the point of consumption, address part of the problem. But dose alone cannot overcome the fundamental strain-mismatch issue. Delivering 50 billion CFU of a strain that your gut's existing community will reject is more effective than delivering 1 billion CFU of the same strain, but it still fails to establish lasting colonisation if the ecological niche is absent.
What Metagenomic Testing Reveals
Shotgun metagenomic sequencing analyses the total DNA extracted from a stool sample, identifying all bacterial, archaeal, and fungal species present in the gut microbiome. Unlike 16S rRNA gene sequencing, which targets a single bacterial gene and can only identify organisms at the genus level, shotgun sequencing reads the entire genome of every organism in the sample. This provides species-level and strain-level resolution across 300 or more bacterial taxa.
The resulting data reveals which bacterial species and functional pathways are present, which are absent, and which are overrepresented. When mapped to metabolic models such as AGORA2 (Assembly of Gut Organisms through Reconstruction and Analysis), a genome-scale metabolic reconstruction database containing 7,302 microbial strains developed by Heinken et al., the sequencing data can predict which introduced strains are likely to find an available ecological niche and which will be rejected by colonisation resistance.
This is the basis of personalised probiotic formulation. Rather than selecting strains from a fixed catalogue of 10 to 15 common species, a metagenomic-guided approach identifies the specific gaps in an individual's microbiome and selects strains that are computationally predicted to fill those gaps. The modelling process uses flux balance analysis (FBA), a mathematical method for simulating metabolic fluxes through biochemical networks, to test whether a candidate strain can sustain growth given the nutrient environment created by the existing community.
Personalised Probiotics vs Generic Supplements
The following comparison outlines the differences between conventional probiotic supplements and metagenomic-guided personalised formulations.
Strain selection
Dose
Verification
Regulatory standard
What the Evidence Shows for Personalised Approaches
Mullish et al. (Neurogastroenterology and Motility, 2024;36(8):e14820) discussed the concept of targeted probiotics designed to restore the balance of specific bacterial taxa rather than supplementing with broad-spectrum formulations. The authors noted that interventions matched to individual microbial profiles showed greater consistency in outcomes compared to universal probiotic supplementation.
GutGutGoose, an Australian personalised probiotics company, uses a Hybrid Mechanistic-AI Framework combining AGORA2 flux balance analysis with machine learning to select strains. Preliminary alpha testing with 20 participants showed 14 times colonisation improvement over baseline when using metagenomic-guided formulations versus the participants' prior generic probiotic regimens, with peer-reviewed publication in preparation. The scientific advisory team includes Dr Robin Mihrshahi of the Sir William Dunn School of Pathology at the University of Oxford.
These early results require validation through larger controlled trials. The 14 times figure is a comparison against participants' own baseline colonisation data using their previous generic probiotics, not a head-to-head randomised controlled trial against a specific competing product. Replication with larger sample sizes and independent research groups will determine whether these results generalise across diverse populations.
Limitations and Considerations
Personalised probiotics address the strain-mismatch problem but do not eliminate all sources of probiotic failure. Diet, medication use (particularly antibiotics), sleep, stress, and other lifestyle factors continue to influence gut microbiome composition and may affect colonisation outcomes regardless of strain selection.
The field of personalised microbiome therapeutics is young. Regulatory frameworks are still adapting to metagenomic-guided interventions, and long-term outcome data beyond 12 to 24 months remains limited across the industry. Consumers should view personalised probiotics as a more targeted approach within a broader strategy for gut health, not as a guaranteed solution in isolation.
Cost is also a factor. Personalised probiotic subscriptions, including metagenomic sequencing and custom compounding, are priced significantly higher than retail supplements. Whether the improved colonisation rates justify the cost difference depends on the individual's health priorities and the severity of their symptoms.
Five Questions People Ask About Probiotic Failure
Why do probiotics make my bloating worse?
Bloating after starting a probiotic can occur when the supplemented strains ferment dietary substrates in sections of the gut where the resident microbiome does not normally produce gas. If the introduced strains are metabolically active but ecologically incompatible with the existing community, they may produce short-chain fatty acids and gas in the small intestine rather than the colon, causing distension and discomfort. This is more common with high-FODMAP diets and with probiotics containing strains that are not matched to the individual's microbial profile.
How long should I try a probiotic before deciding it does not work?
Most clinical trials evaluating probiotic efficacy use intervention periods of 4 to 12 weeks. If no improvement in symptoms is observed after 8 weeks of consistent daily use at the recommended dose, the probiotic strains are unlikely to be colonising. Continuing beyond 12 weeks with the same formulation is unlikely to produce different results, as colonisation either occurs within the first several weeks or does not occur at all.
Can I take multiple probiotic brands at once to improve my chances?
Taking multiple products simultaneously increases the total CFU count but does not address the underlying strain-mismatch problem. If none of the strains in any of the products are compatible with your gut ecology, increasing the volume of incompatible strains will not produce colonisation. In some cases, strains from different products may compete with each other, further reducing the likelihood of any single strain establishing.
Are refrigerated probiotics better than shelf-stable ones?
Refrigeration preserves the viability of certain strains that are sensitive to heat and humidity, but viability at the point of ingestion is only one factor in efficacy. A refrigerated probiotic containing strains that are incompatible with your gut microbiome will not colonise any more effectively than a shelf-stable product with compatible strains. The relevance of refrigeration depends on the specific strains used and the storage conditions, not on a general rule.
What is the difference between probiotic survival and probiotic colonisation?
Survival refers to the probiotic bacteria remaining alive through the manufacturing process, storage, stomach acid exposure, and transit to the large intestine. Colonisation refers to those surviving bacteria adhering to the gut mucosa, integrating with the existing microbial community, and establishing a self-sustaining population that persists after supplementation stops. Most commercial probiotics are tested for survival but not for colonisation in individual consumers.
%20(2).png)
.jpg)